Global mRNA Therapeutics Contract Development & Manufacturing Organization Market: By Product, By Indication, By Application, By End Use, By Region & Segmental Insights Trends and Forecast, 2024 – 2034
- Industry: Healthcare
- Report ID: TNR-110-1158
- Number of Pages: 420
- Table/Charts : Yes
- June, 2024
- Base Year : 2024
- No. of Companies : 10+
- No. of Countries : 29
- Views : 10149
- Covid Impact Covered: Yes
- War Impact Covered: Yes
- Formats : PDF, Excel, PPT
An mRNA therapeutics contract development and manufacturing organization (CDMO) specializes in the comprehensive development and large-scale production of mRNA-based therapies and vaccines for pharmaceutical and biotechnology companies. These organizations offer a range of services including mRNA synthesis, purification, formulation, and aseptic filling, ensuring that the mRNA products meet stringent regulatory standards and quality controls.
Leveraging advanced technologies and expertise in mRNA processes, CDMOs facilitate the rapid and efficient production of these cutting-edge therapies, enabling biotech firms to expedite their research and development timelines and bring innovative treatments to market more swiftly. The role of mRNA therapeutics CDMOs has become increasingly vital in the pharmaceutical landscape, particularly highlighted by their pivotal contribution to the mass production of COVID-19 vaccines, demonstrating their critical importance in addressing global health challenges and advancing medical science.
The rising prominence of mRNA therapeutics, particularly highlighted by the rapid development of COVID-19 vaccines, is a significant demand driver for contract development and manufacturing organizations (CDMOs) specializing in this field. The need for efficient, scalable, and high-quality production of mRNA therapeutics has skyrocketed as pharmaceutical and biotech companies recognize the technology’s potential to revolutionize treatments for a range of diseases, including infectious diseases, cancer, and genetic disorders.
CDMOs provide the necessary expertise and infrastructure for mRNA synthesis, purification, and large-scale manufacturing, addressing the urgent requirement for swift product development and deployment. Additionally, the flexibility of mRNA platforms to quickly adapt to emerging pathogens and personalized medicine further fuels this demand, prompting CDMOs to expand capacities, invest in advanced technologies, and maintain stringent regulatory compliance to meet the dynamic needs of the mRNA therapeutics market.
In terms of revenue, the global mRNA therapeutics contract development & manufacturing organization market was worth US$ 4.72 Bn in 2023, anticipated to witness CAGR of 12.2% during 2024 – 2034.

Global mRNA Therapeutics Contract Development & Manufacturing Organization Market Dynamics
Growing Demand for mRNA Vaccines and Therapeutics– The COVID-19 pandemic has showcased the efficacy and speed of mRNA vaccine development, leading to increased interest in mRNA technology for various diseases. This surge in demand drives the need for specialized CDMOs capable of supporting large-scale production and rapid deployment.
Technological Advancements in mRNA Manufacturing– Innovations in mRNA synthesis, purification, and delivery systems are enhancing production efficiency and product stability. CDMOs are investing in cutting-edge technologies to stay competitive, ensuring they can meet the complex requirements of mRNA therapeutics and deliver high-quality products.
Investment in Infrastructure and Capacity Expansion– To meet the growing demand, CDMOs are expanding their manufacturing capacities and upgrading their facilities. This includes building new production lines, incorporating automation, and enhancing quality control measures, enabling them to handle larger volumes and more diverse projects.
Regulatory Compliance and Quality Assurance– The stringent regulatory landscape for mRNA therapeutics necessitates robust compliance and quality assurance protocols. CDMOs must navigate these regulations effectively to ensure their manufacturing processes meet global standards, which is critical for gaining market approval and maintaining trust with clients.
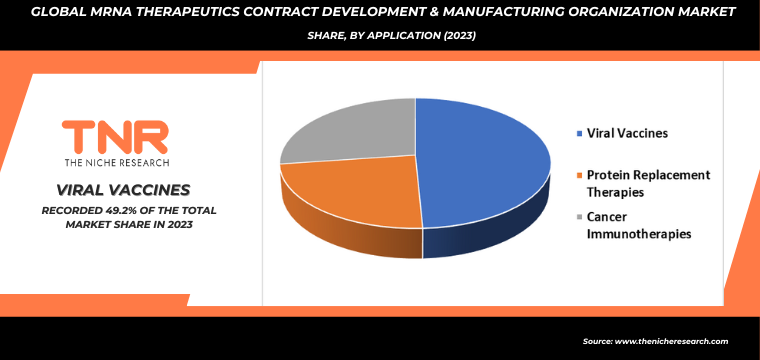
By Indication Infectious Diseases Segment had the Highest Share in the Global mRNA Therapeutics Contract Development & Manufacturing Organization Market in 2023.
The success of mRNA vaccines in combating COVID-19 has demonstrated the technology’s rapid development and deployment capabilities, making it a crucial tool in responding to infectious disease outbreaks. As a result, pharmaceutical and biotech companies are increasingly turning to CDMOs for their specialized expertise in mRNA synthesis, optimization, and large-scale production.
This shift is driven by the need for swift and scalable manufacturing solutions that can meet the urgent demand for vaccines and therapeutics. Additionally, the potential to quickly adapt mRNA platforms to emerging pathogens highlights the necessity for CDMOs with cutting-edge facilities and robust quality control systems. Consequently, CDMOs are expanding their capacities and technological capabilities to cater to the growing and dynamic needs of the mRNA therapeutics market in the realm of infectious diseases.
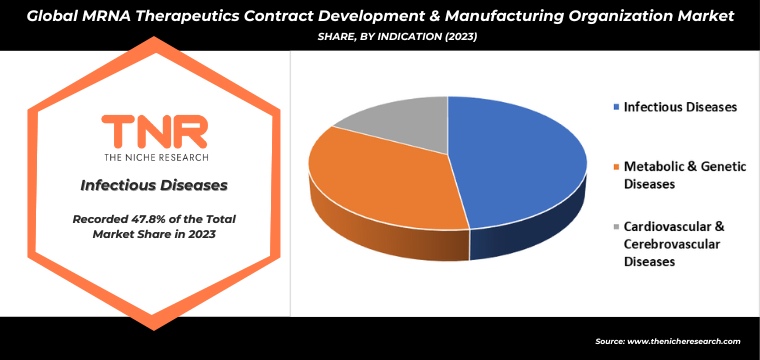
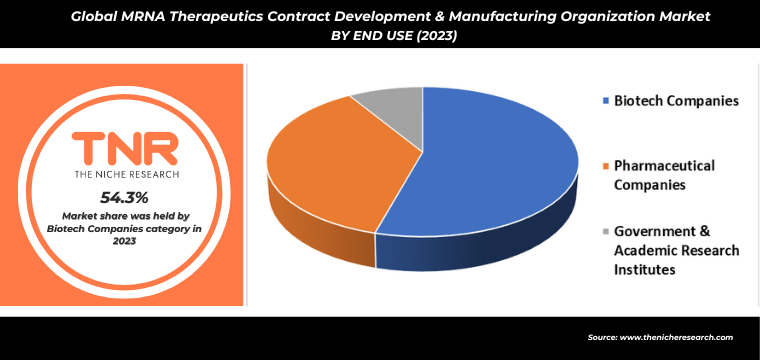
By End Use Biotech Companies had the Highest Share in the Global mRNA Therapeutics Contract Development & Manufacturing Organization Market in 2023.
The escalating interest of biotech companies in mRNA therapeutics is markedly propelling the demand for contract development and manufacturing organizations (CDMOs). The unparalleled success of mRNA-based COVID-19 vaccines has highlighted the technology’s potential, prompting biotech firms to explore its applications across a broader spectrum, including oncology, genetic disorders, and rare diseases. This expanding therapeutic landscape necessitates sophisticated manufacturing processes, pushing biotech companies to partner with CDMOs that offer specialized expertise in mRNA synthesis, purification, and large-scale production.
Additionally, the urgency to expedite time-to-market for innovative treatments underscores the need for CDMOs capable of meeting stringent regulatory standards and ensuring high-quality production. Consequently, CDMOs are witnessing a surge in demand, driving them to invest in advanced technologies and infrastructure to support the dynamic and growing needs of the biotech industry in mRNA therapeutics development.
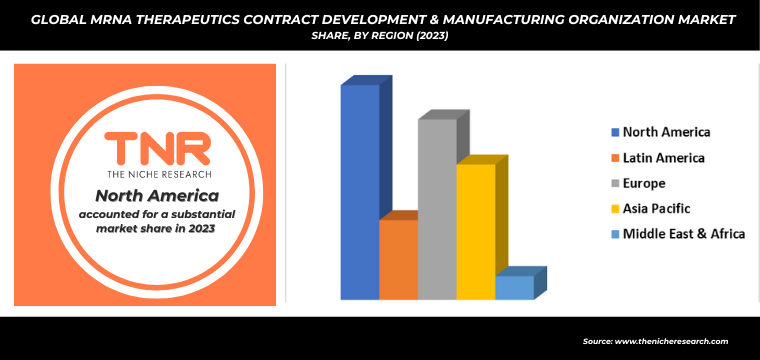
By Region, North America Dominated the Global mRNA Therapeutics Contract Development & Manufacturing Organization Market.
The Growing demand for mRNA therapeutics in North America is significantly driving growth in the contract development and manufacturing organization (CDMO) sector. As mRNA technology proves instrumental in rapid vaccine development, particularly exemplified by the success of COVID-19 vaccines, pharmaceutical companies are increasingly relying on CDMOs for their specialized expertise and scalable manufacturing capabilities. This surge is fueled by the need for quick turnaround times, regulatory compliance, and innovative solutions in mRNA synthesis, purification, and formulation.
Moreover, advancements in personalized medicine and the expanding scope of mRNA applications beyond infectious diseases to include cancer, genetic disorders, and rare diseases further amplify this demand. CDMOs are thus investing heavily in state-of-the-art facilities and technologies to meet the stringent quality and production requirements, positioning themselves as crucial partners in the biotech industry’s pursuit of groundbreaking mRNA-based treatments.
Competitive Landscape: Global mRNA Therapeutics Contract Development & Manufacturing Organization Market:
- ApexBio Technology
- Biocina
- Biomay AG
- BioNTech SE
- Bio-Synthesis, Inc.
- Danaher
- eTheRNA
- Kaneka Eurogentec S.A.
- Lonza
- Novo Holdings
- Recipharm AB
- Samsung Biologics
- TriLink BioTechnologies
- Other Industry Participants
Global mRNA Therapeutics Contract Development & Manufacturing Organization Market Scope
| Report Specifications | Details |
| Market Revenue in 2023 | US$ 4.72 Bn |
| Market Size Forecast by 2034 | US$ 16.74 Bn |
| Growth Rate (CAGR) | 12.2% |
| Historic Data | 2016 – 2022 |
| Base Year for Estimation | 2023 |
| Forecast Period | 2024 – 2034 |
| Report Inclusions | Market Size & Estimates, Market Dynamics, Competitive Scenario, Trends, Growth Factors, Market Determinants, Key Investment Segmentation, Product/Service/Solutions Benchmarking |
| Segments Covered | By Product, By Indication, By Application, By End Use, By Region |
| Regions Covered | North America, Europe, Asia Pacific, Middle East & Africa, Latin America |
| Countries Covered | U.S., Canada, Mexico, Rest of North America, France, The UK, Spain, Germany, Italy, Nordic Countries (Denmark, Finland, Iceland, Sweden, Norway), Benelux Union (Belgium, The Netherlands, Luxembourg), Rest of Europe, China, Japan, India, New Zealand, Australia, South Korea, Southeast Asia (Indonesia, Thailand, Malaysia, Singapore, Rest of Southeast Asia), Rest of Asia Pacific, Saudi Arabia, UAE, Egypt, Kuwait, South Africa, Rest of Middle East & Africa, Brazil, Argentina, Rest of Latin America |
| Key Players | ApexBio Technology, Biocina, Biomay AG, BioNTech SE, Bio-Synthesis, Inc., Danaher, eTheRNA, Kaneka Eurogentec S.A., Lonza, Novo Holdings, Recipharm AB, Samsung Biologics, TriLink BioTechnologies. |
| Customization Scope | Customization allows for the inclusion/modification of content pertaining to geographical regions, countries, and specific market segments. |
| Pricing & Procurement Options | Explore purchase options tailored to your specific research requirements |
| Contact Details | Consult With Our Expert
Japan (Toll-Free): +81 663-386-8111 South Korea (Toll-Free): +82-808- 703-126 Saudi Arabia (Toll-Free): +966 800-850-1643 United Kingdom: +44 753-710-5080 United States: +1 302-232-5106 E-mail: askanexpert@thenicheresearch.com
|
Global mRNA Therapeutics Contract Development & Manufacturing Organization Market
By Indication
- Infectious Diseases
- Metabolic & Genetic Diseases
- Cardiovascular & Cerebrovascular Diseases
By Application
- Viral Vaccines
- Protein Replacement Therapies
- Cancer Immunotherapies
By End Use
- Biotech Companies
- Pharmaceutical Companies
- Government & Academic Research Institutes
By Region
- North America (U.S., Canada, Mexico, Rest of North America)
- Europe (France, The UK, Spain, Germany, Italy, Nordic Countries (Denmark, Finland, Iceland, Sweden, Norway), Benelux Union (Belgium, The Netherlands, Luxembourg), Rest of Europe)
- Asia Pacific (China, Japan, India, New Zealand, Australia, South Korea, Southeast Asia (Indonesia, Thailand, Malaysia, Singapore, Rest of Southeast Asia), Rest of Asia Pacific)
- Middle East & Africa (Saudi Arabia, UAE, Egypt, Kuwait, South Africa, Rest of Middle East & Africa)
- Latin America (Brazil, Argentina, Rest of Latin America)
Report Layout:
Table of Contents
Note: This ToC is tentative and can be changed according to the research study conducted during the course of report completion.
**Exclusive for Multi-User and Enterprise User.
Global mRNA Therapeutics Contract Development & Manufacturing Organization Market
By Indication
- Infectious Diseases
- Metabolic & Genetic Diseases
- Cardiovascular & Cerebrovascular Diseases
By Application
- Viral Vaccines
- Protein Replacement Therapies
- Cancer Immunotherapies
By End Use
- Biotech Companies
- Pharmaceutical Companies
- Government & Academic Research Institutes
By Region
- North America (U.S., Canada, Mexico, Rest of North America)
- Europe (France, The UK, Spain, Germany, Italy, Nordic Countries (Denmark, Finland, Iceland, Sweden, Norway), Benelux Union (Belgium, The Netherlands, Luxembourg), Rest of Europe)
- Asia Pacific (China, Japan, India, New Zealand, Australia, South Korea, Southeast Asia (Indonesia, Thailand, Malaysia, Singapore, Rest of Southeast Asia), Rest of Asia Pacific)
- Middle East & Africa (Saudi Arabia, UAE, Egypt, Kuwait, South Africa, Rest of Middle East & Africa)
- Latin America (Brazil, Argentina, Rest of Latin America)
The Niche Research approach encompasses both primary and secondary research methods to provide comprehensive insights. While primary research is the cornerstone of our studies, we also incorporate secondary research sources such as company annual reports, premium industry databases, press releases, industry journals, and white papers.
Within our primary research, we actively engage with various industry stakeholders, conducting paid interviews and surveys. Our meticulous analysis extends to every market participant in major countries, allowing us to thoroughly examine their portfolios, calculate market shares, and segment revenues.
Our data collection primarily focuses on individual countries within our research scope, enabling us to estimate regional market sizes. Typically, we employ a bottom-up approach, meticulously tracking trends in different countries. We analyze growth drivers, constraints, technological innovations, and opportunities for each country, ultimately arriving at regional figures.Our process begins by examining the growth prospects of each country. Building upon these insights, we project growth and trends for the entire region. Finally, we utilize our proprietary model to refine estimations and forecasts.
Our data validation standards are integral to ensuring the reliability and accuracy of our research findings. Here’s a breakdown of our data validation processes and the stakeholders we engage with during our primary research:
- Supply Side Analysis: We initiate a supply side analysis by directly contacting market participants, through telephonic interviews and questionnaires containing both open-ended and close-ended questions. We gather information on their portfolios, segment revenues, developments, and growth strategies.
- Demand Side Analysis: To gain insights into adoption trends and consumer preferences, we reach out to target customers and users (non-vendors). This information forms a vital part of the qualitative analysis section of our reports, covering market dynamics, adoption trends, consumer behavior, spending patterns, and other related aspects.
- Consultant Insights: We tap into the expertise of our partner consultants from around the world to obtain their unique viewpoints and perspectives. Their insights contribute to a well-rounded understanding of the markets under investigation.
- In-House Validation: To ensure data accuracy and reliability, we conduct cross-validation of data points and information through our in-house team of consultants and utilize advanced data modeling tools for thorough verification.
The forecasts we provide are based on a comprehensive assessment of various factors, including:
- Market Trends and Past Performance (Last Five Years): We accurately analyze market trends and performance data from preceding five years to identify historical patterns and understand the market’s evolution.
- Historical Performance and Growth of Market Participants: We assess the historical performance and growth trajectories of key market participants. This analysis provides insights into the competitive landscape and individual company strategies.
- Market Determinants Impact Analysis (Next Eight Years): We conduct a rigorous analysis of the factors that are projected to influence the market over the next eight years. This includes assessing both internal and external determinants that can shape market dynamics.
- Drivers and Challenges for the Forecast Period:Identify the factors expected to drive market growth during the forecast period, as well as the challenges that the industry may face. This analysis aids in deriving an accurate growth rate projection.
- New Acquisitions, Collaborations, or Partnerships: We keep a close watch on any new acquisitions, collaborations, or partnerships within the industry. These developments can have a significant impact on market dynamics and competitiveness.
- Macro and Micro Factors Analysis:A thorough examination of both macro-level factors (e.g., economic trends, regulatory changes) and micro-level factors (e.g., technological advancements, consumer preferences) that may influence the market during the forecast period.
- End-User Sentiment Analysis: To understand the market from the end-user perspective, we conduct sentiment analysis. This involves assessing the sentiment, preferences, and feedback of the end-users, which can provide valuable insights into market trends.
- Perspective of Primary Participants: Insights gathered directly from primary research participants play a crucial role in shaping our forecasts. Their perspectives and experiences provide valuable qualitative data.
- Year-on-Year Growth Trend: We utilize a year-on-year growth trend based on historical market growth and expected future trends. This helps in formulating our growth projections, aligning them with the market’s historical performance.
Research process adopted by TNR involves multiple stages, including data collection, validation, quality checks, and presentation. It’s crucial that the data and information we provide add value to your existing market understanding and expertise. We have also established partnerships with business consulting, research, and survey organizations across regions and globally to collaborate on regional analysis and data validation, ensuring the highest level of accuracy and reliability in our reports.









